In the relentless drive for fuel efficiency and sustainability, the automotive industry has increasingly turned to lightweight materials. Aluminum alloys are at the forefront of this revolution, offering an excellent strength-to-weight ratio. But what makes this even more compelling is the ability to use recycled aluminum, which requires only a fraction of the energy needed for primary production.
However, using recycled materials isn't as simple as just melting and recasting. Recycled aluminum, often designated as a "secondary" alloy, comes with a more complex chemistry, including higher levels of elements like iron and copper. To unlock its full potential for demanding automotive components like engine blocks or transmission housings, it needs to be carefully processed.
A 2011 study by E. Tillová and her colleagues, published in the Journal of Achievements in Materials and Manufacturing Engineering, dives deep into this challenge. They conducted a detailed structural analysis of a heat-treated, recycled AlSi9Cu3 cast alloy—a true workhorse in the automotive world—to find the perfect recipe for maximizing its performance. Let's break down what they found.
The Challenge: Taming a Complex Microstructure
The alloy in question, AlSi9Cu3, is primarily composed of Aluminum (Al), Silicon (Si), and Copper (Cu). In its as-cast state, its internal structure, or microstructure, is far from ideal. It contains several key features:
- α-Aluminum Dendrites: The primary aluminum matrix that forms the bulk of the alloy.
- Eutectic Silicon: Silicon particles that solidify alongside the aluminum. In their natural state, they form sharp, needle-like platelets that act as stress concentration points, making the material brittle.
- Intermetallic Phases: Complex particles formed by other elements. In this recycled alloy, the most important are:
- Copper-rich phases (e.g., Al₂Cu): These are essential for strength. Heat treatment can dissolve and then re-precipitate these particles in a very fine form, which significantly hardens the alloy.
- Iron-rich phases (e.g., Al₅FeSi): Iron is a common impurity in recycled aluminum. It forms large, brittle needle-like phases (known as β-phase) that are highly detrimental to mechanical properties, especially ductility.
The goal of heat treatment is to manipulate this complex microstructure—to reshape the bad, dissolve and reform the good, and ultimately produce a stronger, tougher material.
The Experiment: The "T4" Heat Treatment Recipe
The researchers applied a T4 heat treatment to the cast alloy. This is a three-step process:
- Solution Treatment: The alloy is heated to a high temperature (just below its melting point) and held there. The team tested three temperatures (505°C, 515°C, and 525°C) for various times (2, 4, 8, 16, and 32 hours). The goal is to dissolve the strengthening copper phases into the aluminum matrix and begin reshaping the silicon particles.
- Quenching: The material is rapidly cooled in warm water. This "freezes" the dissolved elements in place, creating a supersaturated solid solution.
- Natural Aging: The quenched material is left at room temperature for 24 hours. During this time, the dissolved atoms (mainly copper) begin to precipitate out of the matrix as extremely fine particles, which impede dislocation movement and dramatically increase the alloy's strength and hardness.
After treatment, the team performed tensile tests and Brinell hardness measurements and used optical and scanning electron microscopy (SEM) to see exactly how the microstructure had changed.
Key Findings: Finding the "Sweet Spot"
The results provided a clear roadmap for optimizing this alloy.
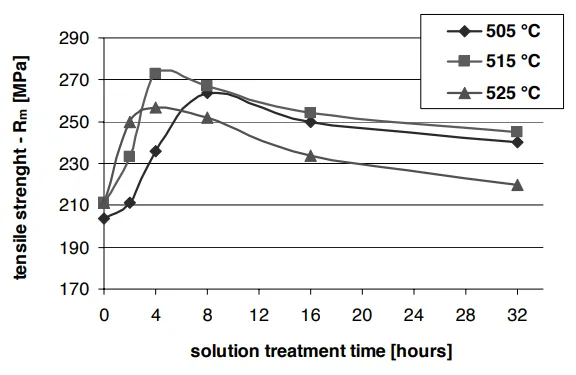
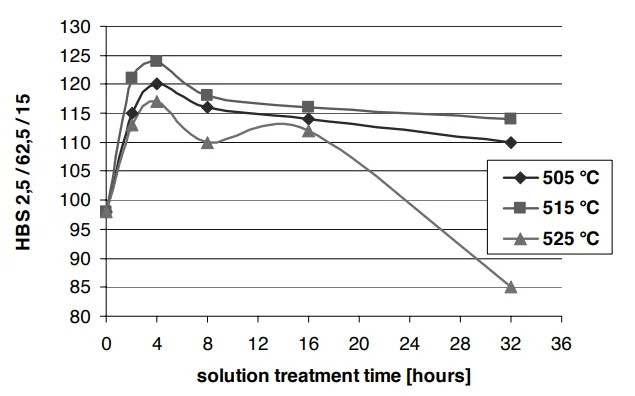
1. Peak Performance is a Balancing Act
The mechanical properties—tensile strength and hardness—didn't just increase with time and temperature. Instead, they peaked and then declined.
The sweet spot was found to be a solution treatment at 515°C for 4 hours.
Fig. 2 & 3 from the paper show a clear peak in both tensile strength and Brinell hardness after 4 hours of solution treatment at 515°C.
Why? At this point, the beneficial effects (dissolution of copper phases and reshaping of silicon) were maximized. Holding the alloy for longer or at higher temperatures led to detrimental effects like the coarsening of particles, which reduces strength.
2. Eutectic Silicon: From Brittle Needles to Rounded Particles
One of the most significant transformations was in the shape of the eutectic silicon.
- As-Cast: Long, sharp platelets (Fig. 6a).
- After Treatment: The heat treatment caused these platelets to fragment and gradually spheroidize, or become more rounded (Fig. 6b-d).
This change is critical. Rounded particles are much less likely to initiate cracks than sharp ones, significantly improving the alloy's ductility and toughness. The optimal spheroidization occurred around 515°C.
Fig. 6 from the paper illustrates the spheroidization of eutectic silicon particles with increasing solution treatment temperature.
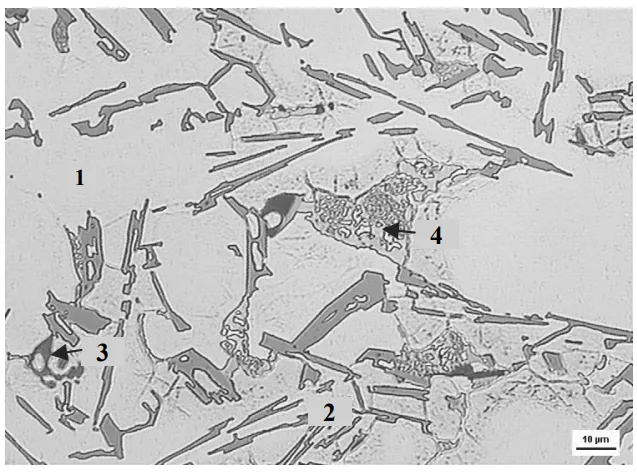
3. Managing the Intermetallics: Dissolution and Danger
Copper Phases: The study confirmed that the solution treatment effectively dissolved the blocky Al-Al₂Cu-Si phases. At 515°C, they broke down and were absorbed into the aluminum matrix, ready to be re-precipitated as strengthening particles during aging.
However, pushing the temperature to 525°C introduced a significant risk: incipient melting. The copper-rich phases have a lower melting point than the bulk alloy. At 525°C, these regions began to melt, creating microscopic voids and structureless phases upon quenching (Fig. 9). This is a primary reason why the mechanical properties dropped off sharply at the highest temperature.
Iron Phases: The researchers observed that the presence of manganese (Mn) in the alloy helped transform the most harmful needle-like iron phase (β-Al₅FeSi) into a more compact, less damaging "Chinese script" or skeleton-like phase (α-Al₁₅(FeMn)₃Si₂). The solution treatment further improved this by causing these α-phase skeletons to fragment and spheroidize, reducing their negative impact even more.
Why This Research Matters
This study provides more than just academic data; it offers a practical guide for the manufacturing world.
- A Precise Recipe: It provides a clear, optimized heat treatment cycle (515°C for 4 hours) for a common recycled automotive alloy, enabling manufacturers to achieve the best possible mechanical properties reliably.
- Highlights Critical Limits: It demonstrates the danger of "over-treating." Pushing the temperature just 10°C too high (to 525°C) can initiate melting and severely compromise the part's integrity. Precise process control is paramount.
- Links Structure to Performance: It beautifully illustrates the fundamental materials science principle that properties are dictated by structure. By visually showing the evolution of silicon, copper, and iron phases, the researchers explain why the strength and hardness change.
- Boosts Sustainability: By defining a robust process for a secondary (recycled) alloy, this research helps make the use of sustainable materials in high-performance applications more viable and reliable.
In conclusion, the work by Tillová et al. is a perfect example of how detailed microstructural analysis can be translated into tangible engineering improvements, helping build the lighter, stronger, and greener cars of the future.
Reference:
[1] C.T. Rios, et al., Intermetallic compounds in the Al-Si-Cu system, Acta Microscopia 12 (2003) 77-82.
[2] S.K. Das, J.A.S. Green, Aluminium industry and climate change - Assessment and responses, JOM 62/2 (2010) 27-31.
[3] G. Mrówka-Nowotnik, J. Sieniawski, Microstructure and mechanical properties of C355.0 cast aluminium alloy, Journal of Achievements in Materials and Manufacturing
Engineering 47/2 (2011) 85-94.
[4] E. Tillová, M. Chalupová, Štruktúrna analýza zliatin Al-Si, EDIS Žilina, Žilina, 2009 (in Slovak).
[5] L.A. DobrzaĔski, R. Maniara, J.H Sokoáowski, The effect of cast Al-Si-Cu alloy solidification rate on alloy thermal characteristics, Journal of Achievements in Materials and Manufacturing Engineering 17/1-2 (2006) 217-220.
[6] J.A. Taylor, The effect of iron in Al-Si casting alloys, Proceedings of the International 35th Australian Foundry Institute National Conference, Adelaide, 2004,148-157.
[7] S. Seifedine, S. Johansson, I. Svensson, The influence of cooling rate and manganese content on the ȕ-Al5FeSi phase formation and mechanical properties of Al-Si- based alloys, Materials Science and Engineering A 490 (2008) 385-390.
[8] A.M. Samuel, F.H. Samuel, H.W. Doty, Observation on the formation ß-Al5FeSi phase in 319 type Al-Si alloys, Journal of Materials Science 31 (1996) 5529-5539.
[9] M.A. Moustafa, Effect of iron content on the formation of ß-Al5FeSi and porosity in Al-Si eutectic alloys, Journal of Materials Processing Technology 209 (2009) 605-610.
[10] M. KrupiĔski, K. Labisz, Z. Rdzawski, M. Pawlyta, Cooling rate and chemical composition influence on structure of Al-Si-Cu alloys, Journal of Achievements in Materials and Manufacturing Engineering 45/1 (2011) 13-22.
[11] E. Tillová, M. Panušková, Effect of solution treatment on intermetallic phase’s morphology in AlSi9Cu3 cast alloy, Materials Engineering 14 (2007) 73-76.
[12] M. Panušková, E. Tillová, M. Chalupová, Relation between mechanical properties and microstructure of cast aluminum alloy AlSi9Cu3, Strength of Materials 1 (2008) 109-112.
[13] E. Tillová, M. Chalupová, M. Panušková, Structural analyses of the Al-Si-Cu alloys, Material Engineering 13 (2006) 25-30.
[14] F.H. Samuel, Incipient melting of Al5Mg8Si6Cu2 and Al2Cu intermetallics in unmodified and strontium-modified Al-SiCu-Mg (319) alloys during solution heat treatment, Journal of Materials Science 33 (1998) 2283-2297.
[15] E. Tillová, M. Panušková, M. Chalupová, Metallographische analyse von Al-Si-Cu Gusslegierungen, Druckguss-praxis 4 (2007) 108-112.
[16] A.M. Samuel, F.H. Samuel, Effect of alloying elements and dendrite arm spacing on the microstructure and hardness of an Al-Si-Cu-Mg-Fe-Mn (380) aluminium die-casting alloy, Journal of Materials Science 30 (1995) 1698-1708.
[17] M. WierzbiĔska, G. Mrówka-Nowotnik, Identification of phase composition of AlSi5Cu2Mg aluminium alloy in T6 condition, Archives of Materials Science and Engineering 30/2 (2008) 85-88.
[18] M. KrupiĔski, K. Labisz, L.A. DobrzaĔski, Structure investigation of the Al-Si-Cu alloy using derivative thermo analysis, Journal of Achievements in Materials and Manufacturing Engineering 34/1 (2009) 47-54.
[19] R. Maniara, L.A. DobrzaĔski, M. KrupiĔski, J.H. Sokoáowski, The effect of copper concentration on the
microstructure of Al-Si-Cu alloys, Archives of Foundry Engineering 7/2 (2007) 119-124.
[20] K. Labisz, M. KrupiĔski, L.A. DobrzaĔski, Phases morphology and distribution of the Al-Si-Cu alloy, Journal of Achievements in Materials and Manufacturing Engineering 37/2 (2009) 309-316.
[21] E. Tillová, M. Chalupová, Effect of solution treatment on Intermetallic phase’s morphology in AlSi9Cu3 cast alloy, mettalurgija/METABK 47/3 (2008) 133-137.
[22] R. Li, Solution heat treatment of 354 and 355 cast alloys, AFS Transaction 26 (1996) 777-783.
[23] F. Paray, J.E. Gruzleski, Microstructure - mechanical property relationships in a 356 alloy, Part I, Microstructure, Cast Metals 7 (1994) 29-40.
[24] L. Lasa, J.M. Rodriguez-Ibabe, Evolution of the main intermetallic phases in Al-Si-Cu-Mg casting alloys during solution treatment, Journal of Materials Science 39 (2004) 1343-1355.
[25] M.A. Moustafa, F.H. Samuel, H.W. Doty, Effect of solution heat treatment and additives on the microstructure of Al-Si (A413.1) automotive alloys, Journal of Materials Science 38 (2003) 4507-4522.
[26] E. Sjölander, S. Seifeddine, Optimisation of solution treatment of cast Al-Si-Cu alloys, Materials and Design 31 (2010) S44–S49.
[27] P. Skoþovský, E. Tillová, J. Belan, Influence of technological factors on eutectic silicon morphology in Al-Si alloys, Archiwes of Foundry Engineering 9/2 (2009) 169-172.
[28] Guiqing Wang, Xiufang Bian, Weimin Wang, Junyan Zhang, Influence of Cu and minor elements on solution treatment of Al-Si-Cu-Mg cast alloys, Materials Letters 57 (2003) 4083-4087.